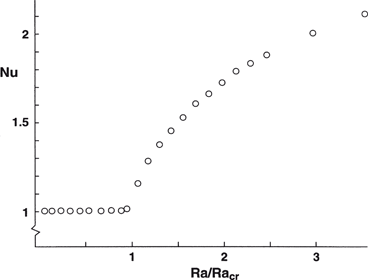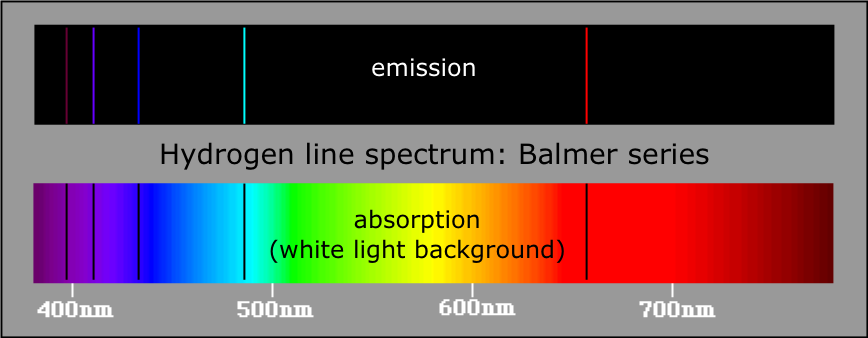
Atomic spectra, simple models of atoms | Introduction to the physics of atoms, molecules and photons

In Bohr's model, for aunielectronic atom, following symbols are used r(n)z rarr Potential energy n+(th) orbit with atomic number Z, U(n,z) rarr Potantial energy of election , K(n,z)rarr Kinetic energy of electron ,

SOLVED: The relative binding of the extra electron in the arsenic atom that replaces an atom in silicon Or germanium can bc understood from calculation of the first Bohr orbit of this

If in Bohr's model, for unielectronic atoms, the following symbols are used, rn,z→ Radius of n^th orbit with atomic number Z ; Un,z→ Potential energy of e^- ; Kn,z→ Kinetic energy of

PDF) Dingle and de Sitter Against the Metaphysicians, or Two Ways to Keep Modern Cosmology Physical | George Gale - Academia.edu

On the basis of Bohr's theory, derive an expression for the radius of the of the n^th orbit of an electron of hydrogen atom.

On the basis of Bohr's theory, derive an expression for the radius of the of the n^th orbit of an electron of hydrogen atom.
Theoretical Modeling and Simulation of Electron-Phonon Scattering Processes in Molecular Electronic Devices Alessio Gagliardi







![download as PDF [9.4MB] - Niels Bohr Institutet - Københavns ... download as PDF [9.4MB] - Niels Bohr Institutet - Københavns ...](https://img.yumpu.com/18272665/1/500x640/download-as-pdf-94mb-niels-bohr-institutet-kbenhavns-.jpg)